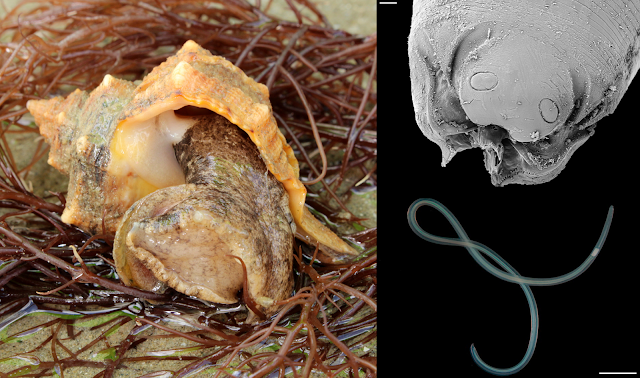
Whales are big animals, even the "smaller" cetaceans such as dolphins and pilot whales are large animals in the range of body sizes across the animal kingdom. The thing about big animals is that they provide a lot of room for parasites. The myriad array of spacious organs found in an animal like a whale provide ample opportunities for parasites to become very specialised not just on a particular species of host, but on a very specific niche within the host's anatomy. Unsurprisingly, there are a wide range of parasites that make their home inside whales. With so many different species inhabiting different parts of the whale anatomy, it is perhaps not surprising that there are worms that specialise in living within the voluminous lungs and sinuses of whales. One of them is a genus named Stenurus.
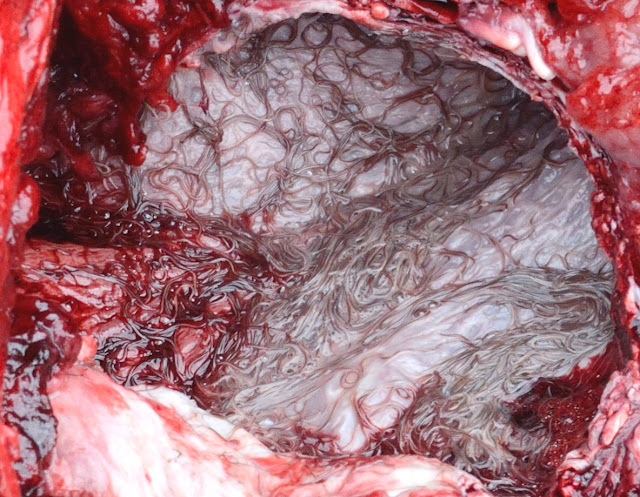 |
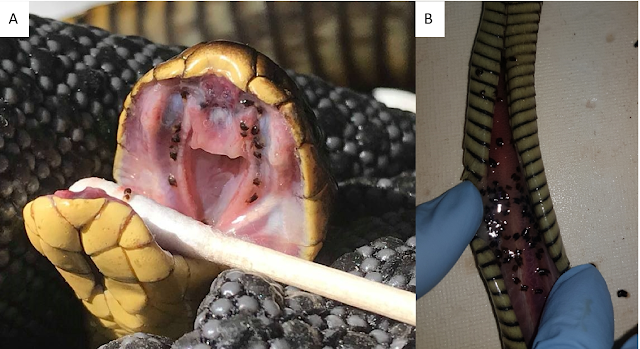
| Numerous Stenurus worms in the pterygoid sinuses of a short-finned pilot whale (Globicephala macrorhynchus). Photo from Fig. 1 of the paper. |
There are nine known species of Stenurus, all of them live in the respiratory system and sinuses of whales. Each of those worms differ in the particular cetacean species that they infect, as well as the part of the host's respiratory system they inhabit. The study being covered in today's blog post were based on samples collected from fourteen toothed whales that had been stranded along the Galicia coast between 2009 and 2019. There were a mix of six different whale species in total, and considering the population of parasites that each whale could support, it provided researchers with plenty of material to work with. Unlike most other parasitological studies where the dissection takes place in the controlled environment of a laboratory, you can't exactly bring a dead whale back to your lab. So instead, the parasites were collected via on-site necropsies.
The researchers found many different species of lung parasites from the different whales, including three species of Stenurus. But out of them, the most abundant was Stenurus globicephalae, which was found in three host species including Risso's Dolphin, Short-finned pilot whales, and Long-finned pilot whales. Each whale harboured between 18 to over 1700 of those worms, which were mostly found in the pterygoid sinus, located deep within the nasal passage near the back of the whale's throat. Previous studies have also recorded S. globicephalae dwelling in the lungs as well as other canals and cavities in a whale's head including the middle ear cavities and cranial sinuses. These worms seem to like dwelling in soft, moist tubes.
Stenurus globicephalae was found to be particularly prolific in short-finned pilot whales, which indicates that while it is capable of infecting other whale species, the short-finned pilot whale just so happens to be a particularly good "fit" for it. Each Stenurus species differ in their host preferences, and it seems to be related to how they get transmitted to the whale in the first place. Like with many other nematodes that infect whales, they do so by hiding in their host's food.
The researchers noticed a certain pattern of association between different species of Stenurus with the diet of the whale species they infect. For example S. globicephalae was associated with species that mostly ate squid, whereas those that ate fish or have a mixed diet tend to be infected with S. ovatus and S. minor. So it is a case of "you are infected by what you eat".
Stenurus was not alone in its preference for whale respiratory structures. In some of the whales, the researchers found Stenurus cohabiting those airy passages with other parasites. One of those co-inhabitants was Halocercus - a different genus of whale lungworm which anchor themselves in place by plunging their head firmly into the host tissue. Another co-inhabitant was Nasitrema - a fluke that lives in the air sinuses of small whales and has a nasty tendency to wander into the host's brain.
While having worms in your sinuses sounds uncomfortable, Stenurus lead a relatively peaceful existence within their wet, cosy homes, and their presence causes surprisingly little to no inflammatory responses. Or at least they do once they settle down as adult worms, because the larval worms can potentially cause focal pneumonia, while the adult worms have a more relaxed relationship with their surroundings. It is as if the whales progressively grow used to their presence, or the worms have grown to tame the fiery response of their host's immune system.
Reference: