This parasite is invisible to the naked eye, can kill its host in 3 days, and it can be found lurking in the waters of rice fields. What I have just described may sound like a nightmare pathogen from a b-horror movie, but it is actually a microscopic flagellated protozoan that has given up a solar-powered life for one fuelled by the blood of its victims. The name of this microscopic monster is Euglenaformis parasitica, and it belongs to a group of otherwise innocuous single-celled critters called Euglenids.
 |
| Left: Scanning electron micrograph of Euglenaformis parasitica. Top Right: E. parasitica extracted from an ostracod host. Bottom Right: E. parasitica visible in the appendage of an infected ostracod. Photos from Fig. 1 and Fig. 7 of the paper |
Euglenids are single-celled flagellated organisms often found in freshwater. The most well-known and well-studied genus is Euglena, which is literally the textbook example of the group, appearing in many biology books as an example of a single-celled eukaryote organism. Many euglenids are photosynthetic, and historically they have been treated as sharing affinity with plants (due to their photosynthetic capabilities) or with animals (due to their active flagellum and being able to take in nutrient via heterotrophy), before getting shunted into a group called the "protist" which is just a jumble of different organisms that scientists couldn't classify into plants, animals, or fungi.
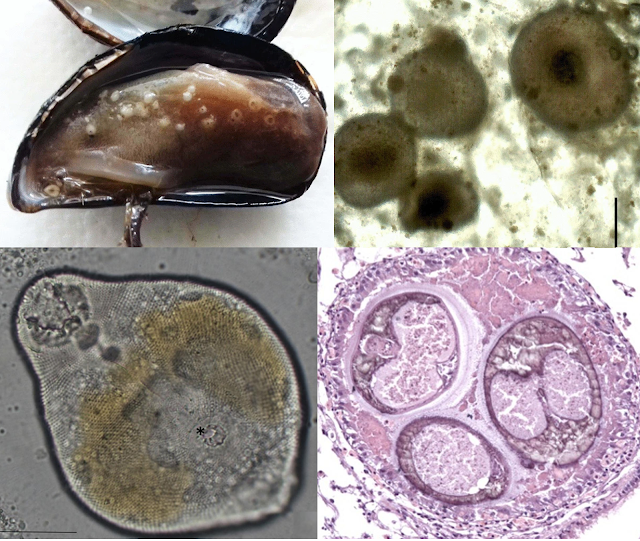
Euglena and its kin are mostly free-living, photosynthesizing when the sun's out, absorbing organic matter from the environment when it's dark. But the ancestor of E. parasitica was not content with this mostly peaceful lifestyle, and has evolved to live inside the body of animals. These euglenids were found parasitising ostracods (also known as seed shrimps) and flatworms in a rice field in Ibaraki, Japan. Ostracods and flatworms belong to two entirely different phyla of animals, and most parasites that infect different phyla of host animals do so at different stages of their complex, multi-stage life cycles. But E. parasitica has just a simple life cycle, which makes it quite remarkable that it is able to adapt to the very different internal environments presented by ostracods and flatworms.
When E. parasitica is in an ostracod, it dwells in the body cavity, swimming in the hemolymph and bathing in its nutrients. Whereas in flatworms, since they don't have any body cavities to speak of, E. parasitica lives in the space between the spongy tissue that forms the bulk of a flatworm's body, burrowing between the cells of the parenchymal tissue. But whether it is in an ostracod or a flatworm, once E. parasitica establishes itself in the host's body, it starts absorbing the literal lifeblood of their host, using it to fuel its exponential growth as it divides and conquers from within.
What started with just a single or a few E. parasitica soon turns into a swarm. This is particularly noticeable in flatworms - uninfected flatworms are semi-translucent, but infected flatworms darken in colour as their body becomes filled with brownish to blackish granules which are actually rapidly dividing E. parasitica. The same goes for ostracods as their blood becomes saturated with the parasite's progenies. After three days, the insides of the host are completely consumed by the swarm of E. parasitica, which proceeds to exit into the surrounding water, leaving behind an empty husk.
There are still a lot of mysteries surrounding this flagellated organism, such as how it is able to make use of such radically different hosts as flatworms and seed shrimps, or how it enters the host's body in the first place. Does it somehow bore through the body wall, or perhaps it tricks the host into eating it, and then burrows through the digestive tract to other parts of the body? If so, it won't be the only parasite to use that trick. There are also questions about its evolutionary origin. Euglenaformis parasitica's close relatives are photosynthetic euglenids, so what made it abandon a solar-powered life in favour of living and reproducing in the bodies of small aquatic animals? Understanding that process would provide us with another clue as to how various different organisms ending up following the path of parasitism.
Reference:
Kato, K., Yahata, K., & Nakayama, T. (2023). Taxonomy of a New Parasitic Euglenid, Euglenaformis parasitica sp. nov.(Euglenales, Euglenaceae) in Ostracods and Rhabdocoels. Protist 174: 125967.